 
Insoluble in water, soluble in ethanol, ether, acetone, chloroform, carbon tetrachloride and benzene. Dodecane is a colorless liquid with a melting point of -9.6☌, a boiling point of 216.3☌, a flash point of 71☌, a density of 0.753g/cm3, and a vapor pressure of 0.133kPa/47.8☌. It may not have been reviewed by professional editors (see full disclaimer).Dodecane is an organic compound with a chemical formula of C12H26 and a molecular weight of 170.33. 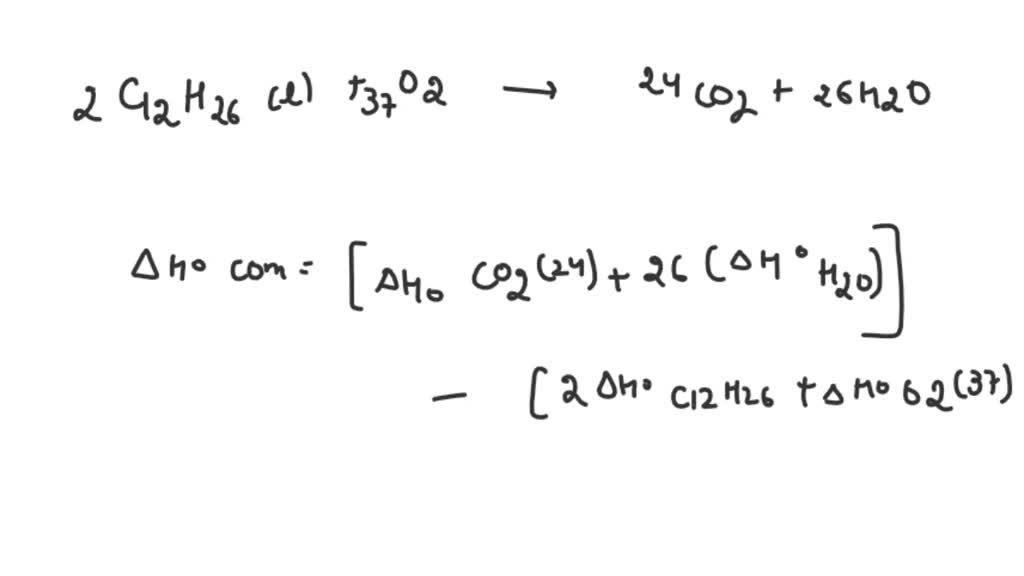
This entry is from Wikipedia, the leading user-contributed encyclopedia. Note that there is no difference between the lower and higher heating values for the combustion of carbon, carbon monoxide and sulfur since no water is formed in combusting those substances. Lower heating value for some organic compounds (at 15.4☌) Fuel Heat of Combustion for some common fuels (higher value) Of some less common fuels ( NIST Chemistry). Heat of Combustion Tables Higher (HHV) and Lower (LHV) Heating values MAF (Moisture and Ash Free) or DAF (Dry and Ash Free) indicates that the fuel heating value has been measured in the absence of inherent moisture and ash forming minerals.MF (Moisture Free) or Dry indicates that the fuel heating value has been measured after the fuel has been dried of all inherent moisture but still retaining its ash forming minerals.AR (As Received) indicates that the fuel heating value has been measured with all moisture and ash forming minerals present.AR, MF, and MAF are commonly used for indicating the heating values of coal: The gross calorific value is relevant for gas burnt in condensing boilers and power plants with flue gas condensation which condense the water vapor produced by combustion, recovering heat which would otherwise be wasted.īoth HHV and LHV can be expressed in terms of AR (all moisture counted), MF and MAF (only water from combustion of hydrogen). This is particularly relevant for natural gas, whose high hydrogen content produces much water. In such applications, the lower heating value is the applicable measure. Most applications which burn fuel produce water vapor which is not used and thus wasting its heat content. Where hv is the heat of vaporization of water, nH2O,out is the moles of water vaporized and nfuel,in is the number of moles of fuel combusted.( Nazaroff, 2007) A common method of relating HHV to LHV is:.This value is important for fuels like wood or coal, which will usually contain some amount of water prior to burning. Gross heating value (see AR) accounts for water in the exhaust leaving as vapor, and includes liquid water in the fuel prior to combustion. 
The energy required to vaporize the water therefore is not realized as heat.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
